TARIFFS
IEEPA Tariffs Ruled Unconstitutional
White House Prepares to Re‐Package Global Fees
A large portion of the U.S. Tariffs imposed beginning in early 2025 have been deemed unconstitutional by the Supreme Court, which announced Feb. 20 that the White house exceeded powers granted to the President to regulate commerce during national emergencies.
Within hours of the Court ruling, the White House announced a new global tariff of 15% on most imported goods for the next 150 days. This tariff is based on Section 122 of the Trade Act of 1974 which permits a temporary import surcharge. Certain goods are exempted, including goods from Canada and Mexico that are already covered by a separate trade agreement.
...On Feb. 23, the European Parliament halted ratification of the Turnberry Trade deal, The European Union's trade deal with the United States that had been negotiated in July. Trade negotiators from India have canceled a planned trip to Washington and Beijing is seeking more information after the ruling.
The tariffs, which had collected more than $200 billion, were imposed under the International Emergency Economic Powers Act (IEEPA) of 1977. The justices ruled 6‐3 against the administration, finding that the IEEPA does not authorize the President to impose tariffs. While the Court did not mandate any refunds to importers, in dissent Justice Brett Kavanaugh warned that the government may be required to repay levies collected. More than 34 million entries have been subject to the tariffs, affecting more than 300,000 importers.
Several lawsuits seek to preserve importer's recovery claims in case refunds become available. If payments were liquidated‐meaning final payments have been made rather than an estimated deposit‐importers may lose their right to apply for refunds.
The IEEPA tariffs ruled unconstitutional include tariffs designed to pressure countries to stop fentanyl from coming into the U.S.; the Russian oil tariffs; the Brazil tariffs; and the reciprocal tariffs. The President used the IEEPA for leverage in many international trade negotiations and as justification for eliminating the de minimis exemption for goods valued under $800 from China.
Other tariffs that the administration has imposed rest on separate authorities: Section 232 of the Trade Expansion Act of 1962 (national security) and Section 301 of the Trade Act of 1974 (unfair trade practices.) Current Section 232 levies include aluminum, copper, and steel, cars, car parts, and transformers. Section 301 tariffs actions have been used to target trade with China.
FOOD & DRUG ADMINISTRATION
Food Traceability Guidance Issued
Q&A Helps Direct Industry
New draft guidance has been issued by the Food and Drug Administration (FDA) to help manufacturers implement the Food Traceability Rule known as FSMA Section 204 (FSMA 204) The deadline for compliance with the Rule has been extended to July 2028. The draft guidance includes non‐binding recommendations regarding meeting the requirements for FSMA 204 including in the context of farming, shellfish, intracompany shipments, and how to determine if a product is on the Food Traceability List. The draft guidance is open for public comment .
The FDA also finalized the exemption for cottage cheese from the Food Traceability Rule. Cottage cheese had been exempted since 2024. That exemption is now permanent. Cottage cheese was kept out of this regulation because manufacturing processes already address the tracing concerns of FSMA 204.
Ultra‐Processed Foods Gaining Attention
RFK ‘Will Act, But Not Regulate’
Health and Human Services Secretary Robert F. Kennedy Jr. says he will act on a citizen petition regarding ultra‐processed foods. In August, former FDA Commissioner Dr. David A. Kessler submitted a 65‐page petition asking the government to reduce exposure to refined carbohydrates and other ingredients to prevent disease. Kessler served as FDA Commissioner of Food and Drugs from 1990‐97.
The petition targets overconsumption of refined sweeteners, refined flours and starches, added fats, oils, and salt, certain corn products, ... extrusion processing and additives such as azodicarbonamide. It challenges the "generally recognized as safe” or GRAS status for some ingredients, arguing science has outpaced those determinations.
In a February 15 interview aired by CBS' 60 Minutes, Secretary Kennedy said he would act on Kessler's petition through consumer education rather than formal regulation. Critics say education alone may not be sufficient while others argue that the UPF label may conflate enriched foods with junk food. The FDA and USDA have been working since 2025 to develop a uniform definition of ultra‐processed foods.
Not FDA but Still Regarding UPFs…
New Non‐UPF Certification Label Offered:
Non-GMO Project, under the non-profit Food Integrity Collective, is offering a Non-UPF Verified Standard label.
The program evaluates products for formulation, ingredient integrity, and processing limits. Non-nutritive sweeteners are prohibited and added refined sugar is limited. Products must be minimally processed and free of high-impact modifications.
The Non-GMO Project has also published an industry brief, "Understanding Ultraprocessed Foods: a Framework for Action." Interested manufacturers need to apply for certification.
FDA's New Year's Resolutions
Reflect Aims for 2026 Regulations :
The FDA&s Human Foods Program released priority deliverables for 2026 with goals to:
- Replace petroleum-based food dyes with safer alternatives;
- Review food additives;
- Reform food regulation and increase transparency about the U.S. food supply;
- Review nutrient requirements for infant formula;
- Create a front-of-package nutrition label program;
- Expand food processing inspections by incorporating state-run inspections;
- Reduce children's exposure to contaminants in food;
- Address concerns about ultra-processed foods (UPF'‐s) and
- Increase transparency in FDA activities.
The agency said that it will focus on three areas: food chemical safety (additives and ingredients); reducing disease through nutrition (labeling, infant formula, UPFs, sodium and added sugar limits, standards of identity); and microbiological food safety (coordinating FDA and state oversight).
Maybe Colors or Maybe Not
‘No Artificial Colors’ Food Dye Labeling Update
The FDA said on February 5 that it will allow products labeled “no artificial colors” or "no added artificial colors" even if they contain added colorings, provided that the colors are not on a list of certified synthetic (petroleum-based) colorings. The move aims to encourage plant-based colors but has drawn criticism that it may confuse consumers and expose producers to state litigation.
It Says "Gluten Free", But Is It Really?
FDA Seeking Input and Considers Changes to Allergen Warnings
The FDA opened a Request for Information on gluten-free labeling and cross contamination. Public comments are open until March 23. The agency seeks input on adverse reactions related to oats, barley, and rye as well as other labeling concerns for consumers.
In addition, the agency is considering changing allergen labels to make them more specific and scientifically based. A meeting on February 18 between scientists, food industry representatives and patient advocates discussed changing the “may contain” portion of a product's label which is used when there is a slight chance a product contains an allergen. In its place, the agency is exploring establishing a threshold for the likelihood of cross contamination.
Public comments on food allergen thresholds are open through May 19 on Regulations.gov.
Is Government Support of BHA Is Going Stale?
FDA Takes a Fresh Look at Preservative Assailed by Health Advocates
Food preservative butylated hydroxyanisole (BHA) is under review by the FDA. The FDA opened a Request for Information as part of its review of chemicals in the nation's food supply. BHA was added to the Generally Recognized as Safe (GRAS) list in 1958 but health advocates consider BHA a likely carcinogen. The RFI closes on April 13. The agency plans to then assess the safety of the preservatives butylated hydroxytoluene (BHT) and axodicarbonamide which is used to condition dough.
U.S. DEPARTMENT OF AGRICULTURE
FAST-ER FOOD?
USDA Seeks to Speed Meat Processing to Drop Prices
The USDA is proposing two update regulations addressing processing requirements for poultry and pork production, removing procedures the agency says slow production in an effort to lower prices. The public will have 60 days to comment once the proposed rules are made available. The USDA says the changes will speed federal line speeds and remove outdated barriers while maintaining oversight and safety. The proposed rules were not available at the time of this publication.
STATESIDE NEWS
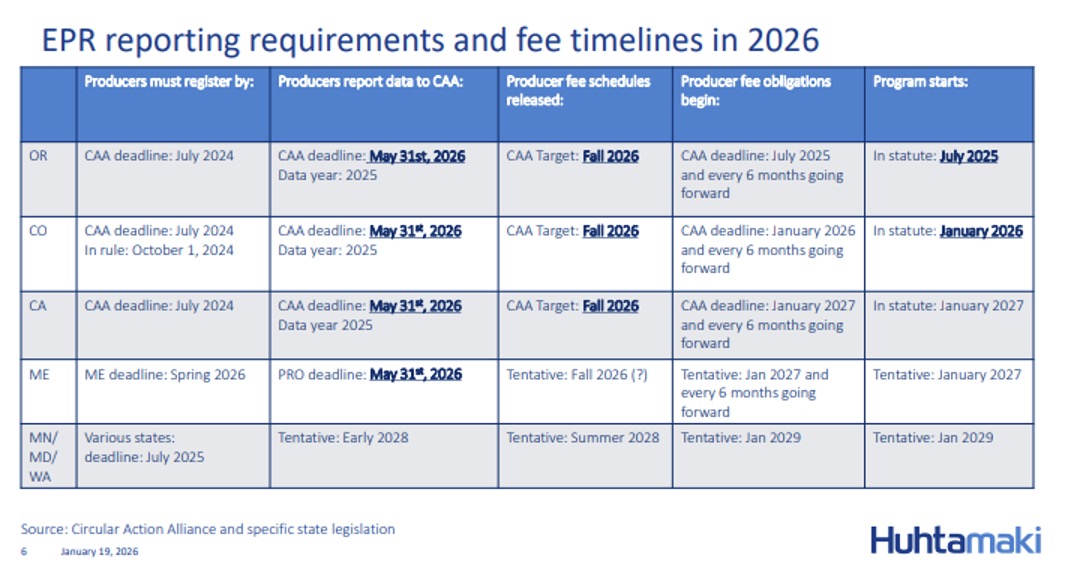
Extended Producer Responsibility Update:
State Recycling Laws In Court
The Circular Action Alliance (CAA) is registering with the Washington Department of Ecology by March 1 to serve as the selected Producer Responsibility Organization (PRO) for members registered by February 15.
CAA is a non-profit alliance of producers guiding compliance with Extended Producer Responsibility (ERP) laws. CAA is acting as a PRO for California, Colorado, Minnesota, and Oregon.
A U.S. District Court in Oregon issued an injunction pausing the enforcement of parts of the Oregon Recycling Modernization Act (RMA) against the National Association of Wholesaler-Distributors (NAW) and its members until a full hearing on the arguments can be held in July. NAW had filed suit in June 2025 challenging Oregon's EPR law.
State Label Laws Halted
Healthier State Agendas vs First Amendment Arguments
A federal court put Texas's “Make Texas Healthy Again” law on hold after a Food & Beverage Industry lawsuit challenged the new warning-label requirements. The law would have required labels by 2027 on foods containing 44 additives stating that those ingredients are “not recommended for human consumption” by other countries. The lawsuit argues that the labels violate the First Amendment because the disclosures are based on the state's opinion and are misleading. A full hearing on the lawsuit is scheduled for this summer.
A similar law banning artificial colors in West Virginia was also blocked. The judge in that case called the ban unconstitutionally vague.
In Texas, a federal court struck down a 2023 law requiring plant-based products marketed as meat to be labeled “meatless” or “lab-grown” in print in equal font size to the product's name. The judge found that the law violated the First Amendment and placed an unjustified burden on producers.
PRESCRIPTION NEWS
Nationwide Pharmacy Benefit Manager Reforms
The Consolidated Appropriations Act, signed into law this month, makes extensive reforms to pass savings to consumers in an attempt to reduce high drug costs. This will apply to patients using Medicare Part D as well as commercial customers.
Pharmacy Benefit Managers (PBMs) must reimburse consumers and insurers quarterly for rebates, and the law severs PBM compensation from being linked to drug prices. PBMs will earn a flat fee for service regardless of the price of the prescription.
PBMs manage prescription drug benefits for insurance companies, employers, and government plans. Major PBMs include CVS Caremark, Optum RX from United Health Group, and Cigna's Express Scripts.
Louisiana 340 B Discount Drug Rule
This month, a federal court rejected challenges to the Louisiana Contract Pharmacy Law which requires drug manufacturers to provide discounted drug pricing under the 340B program to hospitals, clinics and health centers. 340B pharmacies dispense drugs to hospitals and clinics serving low-income or uninsured patients at significant discounts.
POLICIES WE ARE WATCHING
Items below have not changed since our last edition. For details, see previous Policy Watch issues.
• Americans For Ingredient Transparency (AFIT)
In the courts:
• Mondelez vs Aldi
• J.M. Smucker Company vs Trader Joe's Company
• Lululemon Athletica Canada Inc vs Costco Wholesale Corp.
Have feedback or policy you would like us to write about?
Please email our editor, Maureen Donoghue at MDonoghue@PLMA.com
Editor: Maureen Donoghue
MDonoghue@plma.com
